Article
"How do I put it?" – Case reports on well-tolerated HIV post-exposure prophylaxis on deployment in the tropics
From the Department of Tropical Medicine at the Bernhard Nocht Institute (Director: Lieutenant Colonel (MC) Dr D. Wiemer), Bundeswehr Hospital Hamburg (Hospital Commander: Brigadier (MC) Dr J. Hoitz)
Summary
Background: Based on the considerable side effects of first-generation HIV (human immunodeficiency virus) drugs, many medical colleagues consider the need for HIV post-exposure prophylaxis (PEP) to be a reason for the repatriation of military personnel on deployment. However, the improved tolerability of new drugs makes this consideration debatable.
Case report: Based on two case reports about soldiers on deployment in sub-Saharan Africa, successful application of HIV-PEP on deployment after sexual exposure is described. Relevant side effects were not observed. In one instance, HIV-PEP could be aborted after negative testing of the index person. Neither details on the deployments nor on the nationality are presented to protect the anonymity of the affected individuals.
Discussion/Conclusions: Both, the profile of side effects of modern HIV drugs as well as the option of combining them with the currently used anti-malarial drugs allow for the application of HIV-PEP on deployment. Prerequisites are the agreement of the affected one and the availability of a medical role 2 facility to ensure follow-up assessments.
Keywords: HIV, post-exposure prophylaxis, deployment, pre-exposure prophylaxis, follow-up, sexual exposition
"All ignorance is regrettable, but ignorance on so important a matter as sex is a serious danger."
Bertrand Russel, Philosopher
Background
Early post-exposure prophylaxis (PEP), started 2-24 hours or 72 hours at the latest after unprotected sexual contact with an HIV-positive partner, is an important measure of protection from HIV infection. The first generation of HIV medication used as PEP sometimes caused considerable side effects, which in most cases reduced the employability of the affected service members to such an extent that they regularly had to be repatriated. While many still believe that this is the case, the tolerability of drugs available today has improved, as the following case reports show.
Case Reports
Case 1: Celebrating with Bacchus then waking up with Venus and Aphrodite
After intensive counselling by a paramedic who was highly competent, being both a medically qualified confidante and interpreter under national disciplinary control, a serviceman with a good general state of health and nutritional status (approx. 180 cm, 100 kg, bodybuilder figure) presented to the unit physician during consultation hours. His chemo-prophylactic treatment for malaria consisted of 100 mg doxycycline monohydrate daily, which the patient confirmed he was taking regularly. The patient said that he was not taking any other medication.
He reported with obvious shame that he had participated in a bout of drinking one evening with his fellow service members and had consumed a large quantity of alcohol. The next morning he woke up with two African sex workers unable to remember much of what had happened. Apparently, his fellow service members had put some money together and sent the women to his room. Now he was afraid of having become infected with sexually transmitted diseases and was asking to be prescribed post-exposure prophylaxis (PEP). Furthermore, he said amid tears that he was extremely afraid of being repatriated for this reason, fearing not only severe disciplinary but also social implications. His social history revealed that the patient was married and the father of young children. The two sex workers could not be found, which meant that their HIV status could not be determined for the purpose of risk assessment.
The patient was informed in detail about the risks and side effects of PEP, including the significance of "off-label" use of HIV medication, and about the limited diagnostic monitoring options of a medical role-2 facility. Subsequently, PEP was started using a combination of 245 mg/200 mg (1-0-0) tenofovir/emtiricitabine (Truvada®) and 400 mg (1-0-1) raltegravir (Isentress®), stored at the medical facility for this purpose, to be taken for 4 weeks while on deployment. At the start of medication, routine in-theatre lab tests (including liver and kidney parameters) were carried out as zero samples, as well as rapid tests for syphilis and HIV, which were all normal.
The patient returned each week for follow-up examinations. PEP was tolerated well by the subject and medication taken compliantly. The liver and kidney parameters remained normal. After 3 weeks, the unit physician was replaced by a colleague and close contact with the patient was lost. A rapid HIV test carried out as a very early follow-up examination at this time proved negative. The patient was informed that the test was not diagnostically conclusive at that point and the diagnostic window for HIV was extended by the time period needed for PEP.
Case 2: Sexual urge and calculation
A serviceman presented to the unit physician during consultation hours in a role-1 facility in tropical Africa. His prophylactic treatment for malaria consisted of 100 mg doxycycline monohydrate daily, which he took compliantly. The patient reported having had unprotected sexual intercourse with a local woman he met in a bar. He wanted to use a condom but his African partner refused saying that she did not like that. In this situation – giving in to his sexual urge – he complied with her wish but was now concerned about sexually transmitted diseases, especially HIV. He asked to be prescribed PEP and said that for professional and personal reasons a repatriation was not an option for him.
He received detailed information and advice, after which PEP with 245 mg/200 mg (1-0-0) tenofovir/emtiricitabine (Truvada®) and 400 mg (1-0-1) raltegravir (Isentress®) was started. The liver and kidney parameters as well as the results of rapid tests for syphilis and HIV were normal during the first examination at the start of medication.
Seven days after PEP was started, the patient's sexual partner presented to the unit physician during consultation hours for voluntary HIV testing. As his partner's HIV test was negative and her physical examination normal with no indication of seroconversion, the serviceman was able, with the doctor's permission, to discontinue PEP after seven days. When asked about the matter, the woman, who already had a child with an African partner, said that she had a reason for not wanting her partner to use a condom. She wanted to get pregnant by the serviceman in order to escape the difficult social conditions in her country and to be able to migrate to Europe. There were no indications of professional sex work.
Discussion
Risk of exposure through sexual contact on tropical missions
"Prepare your young service members for contact with sexually aggressive cultures." With this urgent warning, Dr Gunther von Laer, former Head of the Health Service of the German Federal Foreign Office, emphasised the importance of prevention of sexually transmitted diseases (STD) on tropical missions as early as September 2003 during an advanced training course at the Bundeswehr Hospital Berlin. In 2006, during the EUFOR RD Congo mission (European Union Force Democratic Republic of Congo mission to provide support during the general elections), his warning was to become even more relevant as large numbers of European-African couples formed on the beaches of Gabon. Encountering a culture in which sexual contact is far more accessible than in comparatively conservative Western and Central Europe and in which the lines between consensual sex and sex work are sometimes blurred constitutes a not insignificant temptation for our service members. This phenomenon of blurred lines can be clearly seen in Case 2, where it was revealed during the anamnesis that the woman had had sexual intercourse with the serviceman not so much because she was attracted to him but because she hoped to be able to migrate if she fell pregnant.
Sexual adventures away from home are highly appealing. A British urology department documented in 1995 that 25% of its patients had had sexual relations while travelling abroad. In two thirds of the cases, condoms were used either not at all or only occasionally [1]. In contrast, since the 1970s German sexual medicine outpatient clinics have been reporting a shift from orgasm disorders towards sexual arousal disorder among heterosexual patients, in other words a lack of sexual desire despite the ability to achieve orgasm. About 60% of female and up to 15% of male patients are affected [2]. The primary reason given for a decline in libido is monotony, boredom, weariness and lack of interest in sexual relations with the same partner [3]. This explains the considerable appeal of a socio-cultural environment in which promiscuous, varied sex is not only fantasised about but can also be experienced, far away from the social controls of home, such as family or friends.
Greater permissiveness is inevitably linked, however, to an increased risk of transmitting STDs. Recently, Sing [4] correctly pointed out that transmission of an STD requires at least three people, parallel or in succession, since a person must have acquired an STD from someone else before he or she can pass it on to another partner. Greater heterosexual permissiveness and promiscuity in sub-Saharan Africa means that heterosexuality in this region is also becoming more relevant to the epidemiology of less infectious sexually transmitted diseases such as HIV. This can also be seen from the gender ratio. While in Europe about 30% of all persons infected with HIV are female, in sub-Saharan Africa it is more than 60% [5].
In sub-Saharan Africa, professional sex work constitutes an additional risk factor, which means that, as described elsewhere [6], single to double-digit percentages of HIV prevalence must be expected in some regions (compared to 0.1% in Germany). Often, HIV prevalence in sex workers is many times higher than in the normal population. Therefore, the contact with two sex workers described in Case 1, if unprotected intercourse did indeed occur while the subject was intoxicated, must certainly be considered relevant to the risk assessment. It should be mentioned that the extreme difference in HIV prevalence between sex workers and the general population cannot be found in Germany. The KABP-Surv STI study on sex workers in Germany from 2010 to 2011[1], conducted by the Robert Koch Institute (RKI) and published on its website (www.rki.de), merely shows an HIV prevalence of 0.2% in sex workers in Germany. This is about twice as much as the German national average.
In view of considerable inhibitions about seeking medical assistance after sexual contact with the risk of infection as described in Case 1, it is important that the physician is perceived as a trustworthy contact who can give immediate and competent advice in real or perceived risk situations and who will take the necessary medical steps.
Risk of HIV transmission through sexual contact
Epidemiological data shows that the HIV transmission risk for one-time sexual contact between serodiscordant but otherwise healthy partners is low [7]. There are minor differences concerning sexual practices (Table 1).
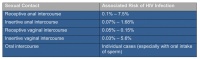 Table 1: Sexual practices and associated risk of HIV infection during one-time sexual contact with an HIV-positive partner [14]
Table 1: Sexual practices and associated risk of HIV infection during one-time sexual contact with an HIV-positive partner [14]
Even sexual practices that are prevalent in some regions of East Africa and associated with a higher risk of contact bleeding, such as "dry sex", for which the vagina is dried out with cloth or paper before intercourse or narrowed and dried by inserting additional objects with the goal of increasing friction during penetration, increase the probability of HIV transmission only to a minor degree, with an odds ratio of less than 1.5 [8].
Nevertheless, the risk of HIV transmission is increased by a factor of 2.5 to 18 [7, 9] if other sexually transmitted diseases are also present, by a factor of 10 to 30 with a moderately increased HIV viral load (< 2500 copies per ml), and even by a factor of 10 to 100 [7] during seroconversion. The probability of transmission during the course of an HIV infection is highest due to the very high viral load during seroconversion and the transition to AIDS (acquired immune deficiency) [10].
Preventive Measures
As stated above, the transmission of STDs requires at least three people, which means that abstinence or fidelity could effectively prevent the spread of disease. Meta-analyses of studies of what are known as "abstinence only" prevention programmes [11, 12] have shown, however, that this approach is counter-productive because it ignores human needs and is therefore unlikely to be adhered to. On the contrary, one of the studies on the "abstinence only" approach showed that the relative risks of unwanted pregnancies had more than doubled to 2.35 and of contracting sexually transmitted diseases to 2.73, compared to the control group. In his work Sing [4] quotes the apt remark of former U.S. Surgeon General Dr Jocelyn Elders: "We know the vows of abstinence break far more easily than latex condoms".
Meta-analyses have indeed shown that condoms reduce the already low risk of HIV transmission by a further 80% on average, which currently provides the best protection available – aside from abstinence and fidelity [13]. As Sing writes in his overview, protection against the transmission of STDs can still be expected even if the condom is damaged. Compared to not using condoms, the relative risk of STD transmissions is 0.006 with burst condoms, 0.000008 with visible leaks and merely 0.00000004 with invisible leaks [4]. Nevertheless, for safety reasons condoms should be imported from Germany where controlled quality is ensured and where they are not exposed to damaging influences such as friction, drying out and oily lubricants.
As demonstrated by the case studies described above, there are, however, still plenty of reasons why condoms are not used in practice. In such cases, medical treatment with PEP may be indicated, depending on the type of exposure.
Aside from PEP, the German-Austrian guideline [14] recommends that immediate measures be taken after exposure to HIV through sexual contact as summarised below (Table 2).
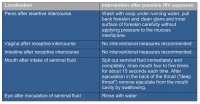 Table 2: Localisation-related immediate measures after possible exposure to HIV through sexual contact [14]
Table 2: Localisation-related immediate measures after possible exposure to HIV through sexual contact [14]
Even if the goal of reducing the viral load with these recommendations is obvious, the relevance of such recommendations seems questionable in practice. Since during consensual unprotected sex there is no immediate expectation of the partner being infected with HIV, concern about a possible exposure to HIV – as described in Case 1 – is likely only after the Dionysian intoxication has worn off. But by that stage it is usually too late to take immediate action. In cases of sexual violence, however, it is doubtful whether the victim, possibly in a state of shock, will be capable of such coordinated action. If appropriate interventions are tolerated, medical staff providing initial treatment would assume a supporting role, while in view of the forensic aspect, considerations regarding protection from infection may be in conflict with those regarding the collection of forensic evidence. Since no evidence-based immediate measures are available for receptive sexual practices that victims of sexual violence typically have to endure and where there is a risk of HIV infection, PEP, as described below, will be the preferred treatment.
Indications for PEP after possible exposure through sexual contact
The current German-Austrian guideline for post-exposure prophylaxis against HIV infections published in 2013 [14] describes differentiated indications for PEP after possible exposure through sexual contact. If a partner is known to be HIV-positive and unprotected vaginal or anal intercourse has occurred, recommendations will be made in accordance with the viral load. For a viral load of > 1000 copies/ml, PEP is recommended, for 50 – 1000 copies/ml it will be offered, whereas successful viral suppression through treatment with a viral load of < 50 copies/ml does not constitute an indication for PEP.
With regard to occasional sexual contact, however, knowing the HIV status of the partner is the exception rather than the rule. If the partner's HIV status is unknown and unprotected intercourse has occurred, recommendations will be much more complex. Table 3 summarises the indications in Germany and Austria for PEP after possible exposure through sexual contact and if the partner's HIV status is unknown.
![Table 3: Indications in Germany and Austria for PEP after possible exposure through sexual contact and unknown HIV status of the partner [14]](/media/article/2967/image-1491486628.jpg)
In the cases described above, PEP indication was justified by the occurrence of vaginal intercourse with partners from the high-prevalence area of sub-Saharan Africa. In Case 1, however, one may justifiably question whether the state of the obviously severely intoxicated serviceman allowed for intercourse with the two sex workers that would have enabled transmission of the virus. The decision was made in favour of the patient's safety, since risk-bearing contact could not be excluded either due to the presumably alcohol-related gap in the patient's memory.
In the case of the second patient, a negative HIV test was obtained from the African sex partner, albeit with seven days' delay, so that the presumed exposure could be identified as a pseudo exposure. PEP was discontinued as a result. This case demonstrates that, if in doubt, PEP can be started but also discontinued if the transmission risk is not confirmed.
As described in the case studies, it is important to inform the patient about "off-label" use of medication and to document this. Even if indication is secured by the guideline valid since 2013 [14], the substances are not formally authorised for this indication.
Table 4 lists HIV prevalences in important Bundeswehr theatres of operation based on MEDINT analyses. They provide a rough guide for estimating the local risk of exposure. Due to mostly fragmentary and unreliable surveillance in crisis areas, such data can only be regarded as approximate values.
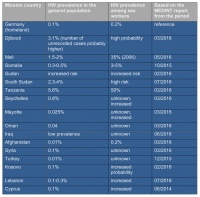 Table 4: HIV prevalences in Bundeswehr theatres of operation compared to the reference standard in Germany
Table 4: HIV prevalences in Bundeswehr theatres of operation compared to the reference standard in Germany
PEP used in accordance with the German-Austrian guideline – can it really be used in the tropics?
The effectiveness of PEP is estimated at 80%, based on a 20-year-old case-control study, which at the time did not include a comparison of zidovudine (Retrovir®) with medication administration after percutaneous exposure [15]. More recent placebo-controlled studies do not exist because such an approach is no longer considered ethical. Thus, there is a lack of evidence to support the actual effectiveness of new substance combinations for PEP. The convention of prescribing PEP for 4 weeks is ultimately based on the opinion of experts. For this reason, the guideline includes the option of prolonging the duration of application according to the individual situation [14] for cases of massive exposure (e.g. through transfusion of infected blood products) or of late initiation of PEP (> 36 hours after exposure).
As described in Case 2, it is correct and important to start PEP at an early stage if there are any doubts [16], because treatment will only be effective if initiated within 72 hours or better still within 2 hours [14]. In the latter case, it is possible to deliver the drug in a targeted manner before HIV becomes integrated in human cells, which in cell cultures takes between 30 minutes and a few hours [17].
An essential prerequisite for PEP effectiveness is also the application of substances against which the potentially transmitted virus has not developed any resistance. In Europe, under conditions of an antiretroviral therapy (ART) with the viral load controlled, resistant viruses are comparatively rare in the case of newly infected individuals [18], while the same certainly cannot be assumed in sub-Saharan Africa where ART is frequently administered without any regular control of the viral load and thus performed "blind", so to speak. A current study from West African Togo [19] provides an example of how, after only 4 years of "blind" ART, 88% of HIV patients showed resistance to nucleoside/nucleotide reverse-transcriptase inhibitors (NRTI) as well as non-nucleoside reverse-transcriptase inhibitors (NNRTI). In this context, it should be noted that until the guideline change in 2013, the Bundeswehr standard PEP in operations abroad consisted of the two NRTI zidovudine and lamivudine (Combivir®) as well as the NNTRI efavirenz (Sustiva®) so that a high risk of failure was likely. For future missions it is recommended that local resistance to HIV be determined by the MEDINT capability of the Bundeswehr Medical Service and that the deployment-specific composition of PEP be adapted accordingly, accompanied if necessary by active surveillance based on resistance testing of locally acquired samples if there is no reliable data available.
A combination of the two better tolerated NRTI tenofovir/emtiricitabine (Truvada®) and the integrase inhibitor raltegravir (Isentress®), recommended by the German-Austrian guideline [14] as standard PEP for Germany and Austria, was selected primarily due to the good tolerability of these medicines [20]. Since the new and, for this reason, expensive integrase inhibitor raltegravir is not used in "blind" ART schemes in sub-Saharan Africa, the integrase inhibitor that rapidly decreases the viral load is likely to remain effective. Nevertheless, an estimate of effectiveness based on speculations remains unsatisfactory considering the consequences of an HIV infection and high rates of virological resistance to be expected in sub-Saharan Africa [19, 21, 22]. For situations of known or expected resistance, the guideline [14] also includes the option of deviating from standard PEP. Such an option should be discussed with experts in infectiology, if possible.
Tolerability of standard PEP and deployment-related medication interaction risks
The current standard PEP Truvada®/Isentress® is characterised by good tolerability, which a study has confirmed [20]. This was also an essential reason for the decision, in the cases described, to use PEP on deployment. The side effects described in this study were generally mild and self-limiting with no cases of PEP termination attributed to side effects. More specifically, the observed side effects were limited to symptoms which, in comparison with the consequences of an HIV infection, can be regarded as minor complaints, e.g. nausea and vomiting (27%), diarrhoea (21%), headache (15%), fatigue (14%), gastrointestinal symptoms (pain, swollen abdomen, flatulence) (16%) as well as myalgia and arthralgia (8%). Serious side effects caused by Travada®, which require a change of medication, such as increased creatine kinase levels (CK), nephrotoxicity and pancreatitis [14], rarely occur and are hardly to be expected if the drug is taken only for a short time during PEP. Raltegravir-associated hepatotoxicity can be controlled in most cases [14].
On tropical missions where malaria prophylaxis is essential, the potential interaction with the anti-malarial drugs taken is always to be observed (Table 5).
The medical checks recommended in Table 5 can be performed without any difficulties even under the limited diagnostic conditions posed by a medical role-2 facility as described in Case 1. In Case 2, follow-up examinations were not required because PEP could be discontinued after a few days as it was very likely that the sexual partner was not infected with HIV. In the cases described, the use of doxycycline for malaria prophylaxis makes checks during PEP relatively simple. However, if the patient is taking mefloquine for malaria prophylaxis, which involves an increased risk of drug interaction, a change of medication may be considered, but attention must be paid to the slow elimination kinetics and associated long half-life of mefloquine.
The recommendation to avoid boosters, such as ritonavir and cobicistat, in combination with all anti-malarial drugs becomes relevant if, in cases of known or expected resistance, boosted protease inhibitors associated with a high resistance barrier are to be used. The guideline [14] specifies, for example, ritonavir-boosted lopinavir (Kaletra®) as a substitute for Isentress, which penetrates the placental barrier. This, however, is of less importance on military operations since pregnant women are repatriated anyway for the protection of their unborn child. Table 6 provides an overview of substances which play or have played a role in PEP use.
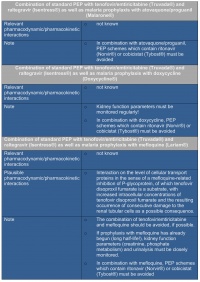 Table 5: Tolerability of malaria prophylaxis together with the current standard PEP (research by Assistant Professor Dr Nils von Heutig, specialist for clinical pharmacology, Frankfurt-Sachsenhausen/Institute for Clinical Pharmacology at the Johann W. von Goethe University, Frankfurt/Main)
Table 5: Tolerability of malaria prophylaxis together with the current standard PEP (research by Assistant Professor Dr Nils von Heutig, specialist for clinical pharmacology, Frankfurt-Sachsenhausen/Institute for Clinical Pharmacology at the Johann W. von Goethe University, Frankfurt/Main)
Interactions with other accompanying or life-style drugs will not be discussed in detail at this point but should be taken into consideration on a case-by-case basis. Muscle-building substances are worth mentioning in this context. Despite his bodybuilder figure, the serviceman described in Case 1 denied taking such substances. If possible, such accompanying medication should be interrupted or discontinued and, in weighing up the risks and benefits to the patient, it should be explained to the patient that the effectiveness of an antiretroviral medication is of utmost importance.
A glimpse into the crystal ball: PrEP and malaria prophylaxis?
Most recently, what is known as pre-exposure prophylaxis (PrEP) is being discussed as a means of cost-effective prevention for persons with risky sexual behaviour for whom using a condom is not an option due to lifestyle considerations. The recently published IPERGAY[1] study about "PrEP on demand" demonstrated a significant as well as quantitatively relevant reduction of the sexual HIV transmission risk [23]. The medication used was Truvada®, which – as described in detail elsewhere [24] – accumulates in large concentrations in genital secretions. For PrEP on demand, 2 tablets of Truvada® are taken with a meal 2-24 hours before the expected sexual risk contact, a further tablet 24 hours after this initial "loading dose" and a fourth tablet 24 hours after that. Therefore, with four tablets of Truvada®, corresponding to a price of just over 100 euros, which is expected to fall dramatically as the patent will expire soon, protection from HIV transmission can be achieved that may not be completely safe but is considerably effective. The few breakthrough infections observed during the IPERGAY study were mostly due to insufficient drug levels, i.e. lack of compliance, and only in exceptional cases due to a real virological failure [23].
 Table 6: Main advantages and disadvantages of HIV medication which play or have played a role in PEP
Table 6: Main advantages and disadvantages of HIV medication which play or have played a role in PEP
Even if CDC[2] and EACS[3] guidelines already recommend PrEP for risk collectives (Table 7), statutory health insurance does not yet cover the costs, which means that this option for the time being remains a "luxury" for the financially better-off. Nevertheless, due to the proven cost-effectiveness as a prophylactic measure for risk groups [25], an evaluation is to be expected for the German guidelines and may influence the financial decisions of health-insurance companies in the near future. If, in justified individual cases, insurance companies decide to pay for PrEP, the Bundeswehr Medical Service will also have to make its position clear on this issue. Irrespective of the obstacle of cost coverage by statutory health insurance, the Bundeswehr Medical Service could, and indeed should, clarify its position on the topic of PrEP. In principle, a Truvada®-based PrEP could be combined with current standard malaria prophylaxis.
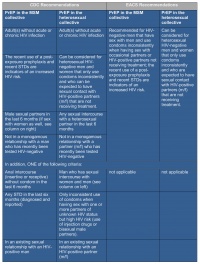 Table 7: Definition of risk collectives for PrEP in accordance with CDC and EACS
Table 7: Definition of risk collectives for PrEP in accordance with CDC and EACS
Social and ethical implications
For the patient, repatriation due to PEP may result in consequences that go far beyond disciplinary matters, as depicted in Case 2. The serviceman described in Case 2 voiced considerable fears about losing his family if his wife were to learn why he had returned home early from deployment. Much like STD-prevention programmes that do not accept that, despite evidence to the contrary [11, 12], concepts of abstinence do not reflect the reality of human life, the same applies to conservative marriage and relationship models in our society. Treating human needs as taboo on account of such conventions leaves the affected persons with no option other than lying. Alternative, open relationship models, such as the concept of "social fidelity" rather than "sexual fidelity" presented by sex educator Oswald Kolle ("Leben 2034: Die Zukunft der Sexualität", Körber-Forum Hamburg, 25 March 2009), have so far met with little acceptance by society, even if they more accurately reflect the reality of life for people who frequently have to work abroad.
The commitment of the paramedic in Case 1, who presented the affected patient, almost against his will, for post-exposure medical care, is to be emphasised and commended as an example of comradeship and humanity. This should certainly not be taken for granted. Even if, under the law, sex between two or more consenting adults is in principle their own business, uninvolved persons may be expected to have negative feelings such as jealousy or envy. The problems that arise when individuals are involved to differing degrees in sexual activity, possibly leading to feelings of rejection and failure, have been satirised in the novel "Extension du domaine de la lutte" by controversial writer Michel Houellebecq.
Medical ethics, however, should follow the four principles of "patient autonomy", "beneficence", "non-maleficence", and "justice" [26]. Therefore, repatriation as a personnel measure must never be used for disciplining the individual concerned. In accordance with this principle, options were sought in the cases described that would enable the affected patients to receive appropriate medical treatment while on deployment.
Finally, it is important to remember that the Bundeswehr has been transformed from a national defence force into an army on operations. This means that sometimes longer deployments abroad will, in future, be the rule rather than the exception for military personnel. Military personnel are required to spend a considerable part of their lives under deployment conditions with risk to life and limb. It is therefore inconsistent and, moreover, in contradiction to earlier epidemiological data [11, 12] to expect them to spend these relevant parts of their lives in abstinence and – despite an uncertain future – refrain from sex as an essential element that creates happiness and gives meaning to life.
There is no medical evidence to support any distinction between a "healthy monogamous" and an "unhealthy promiscuous" sex life as long as STDs, with sometimes chronic symptoms, and unwanted pregnancies, with their psycho-social implications, are avoided by taking suitable preventive measures or their consequences mitigated. Discrimination on the basis of sexual orientation is also prohibited by the General Act on Equal Treatment, with which Anti-Discrimination Directives of the EU were incorporated into German law.
 Table 8: Regulations of NATO partners USA, UK and France regarding PEP use on missions abroad (source: NATO Deployment Health Surveillance Capability, Munich; as of August 2016)
Table 8: Regulations of NATO partners USA, UK and France regarding PEP use on missions abroad (source: NATO Deployment Health Surveillance Capability, Munich; as of August 2016)
It is left to the discretion and responsibility of the individual to decide how he or she satisfies his/her sexual needs within the limits of the law, including while on deployment. In accordance with the above-mentioned principles of "patient autonomy" and "beneficence", medical practitioners must assume an unbiased advisory position. For this purpose, the principle of "primum non nocere" (first, do no harm), which developed from the Hippocratic tradition, may serve as a guideline to ensure a balanced consultation that takes into account the individual needs of each patient and respects his freedoms, but which also imparts vital professional knowledge in drawing the patient's attention to the associated risks, as well as attempting to minimise such risks by looking for suitable approaches. Relevant medical expertise is an important prerequisite for this.
Moreover, it remains unquestionably and primarily the task of medical personnel to ensure the operational capability of service members, which includes enhancement (prevention, including counselling corresponding to the individual's needs and the situation at hand), maintenance (entailing, for example, the introduction and use of PrEP) and/or reestablishment (meaning the use of PEP in accordance with current standards). The performance of this task is in line with the principles of medical ethics and also the employer's duty of care, which involves doing everything possible to keep deployed service members healthy so that they are able to fulfil their mission.
Conclusions
To sum up, it can be said that standard PEP, currently available on missions, is characterised by good tolerability [20] and, using simple laboratory tests, can be combined with anti-malarial drugs routinely prescribed on missions. Mefloquine (Lariam®), which is disappearing from the German market, cannot be combined as well with PEP, which is confirmed by the current practice of avoiding using this substance as the medication of first choice for malaria prophylaxis. Co-medication with PEP in addition to malaria prophylaxis should be avoided, if possible, since polypharmacy leads to increasingly complicated interactions between drugs. Therefore, application of PEP is generally also possible during deployment, as is already practised by NATO partners (Table 8), but, of course, assumes the prior consent of the patient to such a procedure. Confidently recognising the need for PEP, in other words, correctly detecting its indication is also a basic prerequisite.
It would be desirable in future to provide MEDINT information about HIV resistance in theatres of operation on the basis of which resistance-adapted PEP combinations could be calculated.
Moreover, the authors believe it is important to propagate an image of the physician that defines him or her as a trustworthy, approachable advisor, including in situations associated with shame and embarrassment, and as someone who will support the patients entrusted to them primarily by providing them with advice and help but also, if necessary, warning them of the risks. In particular, this will ensure that any necessary time-critical medical measures can be started immediately if, for example, a patient needs to be prescribed PEP [14].
In addition to professional expertise and confidence, the medical expert also requires a high degree of empathy in order to be able to handle conversational situations that may involve discussing the patient's feelings of shame and guilt, as well as fear of moral or even real sanctions. This is the only way to provide the patient with comprehensive and targeted support. In this connection, there is a need for improvement particularly in medical training that medical societies have so far been hesitant to address (for example with the workshop "Let's talk about sex (for medical students)" during the 13th Congress for Infectious Diseases and Tropical Medicine (KIT 2016 in Würzburg)). Effective training to handle complex emotional situations would also be highly desirable for medical personnel on missions abroad. This is particularly true as service members on deployment are generally not able to choose the physicians that treat them.
Key Statements
- Unprotected sexual contact in tropical, sub-Saharan Africa are associated with an increased risk of infection with pathogens of sexually transmitted diseases (STD), including the human immunodeficiency virus (HIV).
- In the case of infection-relevant heterosexual contact (vaginal or anal intercourse) with partners of unknown HIV status from tropical regions where HIV is highly endemic, PEP (post-exposure prophylaxis) should be offered, for MSM contact (men who have sex with men), it must be recommended.
- Standard PEP, consisting of tenofovir/emtricitabine (Truvada®) and raltegravir (Isentress®), is characterised by acceptable tolerability and can be combined with anti-malarial drugs prescribed on tropical missions of the Bundeswehr, assuming reasonable monitoring efforts are made.
- After providing detailed information about "off-label" use, and subject to the diagnostic options of a medical role-2 facility for follow-up examinations, PEP is medically justifiable, including under deployment conditions in tropical regions, and without the need for repatriation.
- It would be desirable for MEDINT to provide surveillance data about HIV resistance in the theatres of operation and, if such reliable data is lacking, for it to be generated using laboratory-based methods as early as the deployment/mission planning stage, in order for PEP to be adapted to the respective missions based on this resistance data.
Acknowledgements
The authors would like to thank Assistant Professor Dr Nils von Hentig (Sachsenhäuser Praxis für Allgemeinmedizin Kiana Darab-Kaboly/Nils von Hentig) for his research of the patterns of interaction to be expected between PEP, based on tenofovir/emtricitabine and raltegravir, and the substances atovaquone/proguanil, doxycycline and mefloquine, used for malaria prophylaxis.
Conflicts of interest
The authors declare that there are no conflicts of interest with regard to the guidelines of the International Committee of Medical Journal Editors.
[1] IPERGAY (Intervention Préventive de l'Exposition aux Risques avec et pour les Gays), a work and study group of the Agence Nationale de Recherche sur le Sida et les Hépatites Virales (Institute of the French Ministry of Health)
[2] CDC = Centers for Disease Control and Prevention (https://www.cdc.gov/hiv/pdf/prepguidelines2014.pdf)
[3] EACS = European AIDS Clinical Society (http://www.eacsociety.org/files/guidelines_8.0-english-revised_20160610.pdf)
[1] KABP = Knowledge, Attitude, Behaviour, Practices, Surv = Surveillance, STI = sexually transmitted infections. The study was an integrated biological and behavioural surveillance study of female sex workers in 12 federal states of Germany. (https://www.rki.de/DE/Content/InfAZ/S/STI/Studien/KABPsurvSTI/KABPsurvSTI_inhalt.html).
Literatur
- Hawkes S, Hart GJ, Bletsoe E, Shergold C, Johnson AM: Risk behaviour and STD acquisition in genitourinary clinic attenders who have travelled. Genitourin Med 1995; 71: 351 - 354.
- Hauch M: Lust auf Dissens. Heterosexualität in der De/Re/Konstruktion. In: Dannecker M, Reiche R (Hg.). Sexualität und Gesellschaft. Festschrift für Volkmar Sigusch. Campus Verlag GmbH Frankfurt/Main 2000; 217 - 218.
- Bräutigam W: Sexualmedizin im Grundriß. Ein Einführung in Klinik, Theorie und Therapie der sexuellen Konflikte und Störungen. Georg Thieme Verlag Stuttgart 1977; 214.
- Sing A: Zur Epidemiologie von sexuell übertragbaren Erkrankungen: der Beitrag der Sozialen Netzwerk-Analyse zu einem komplexen Feld. Mikrobiologe 2011; 21: 150 - 159.
- Piot P, Carael M: Global Perspectives on Human Immunodeficiency Virus Infection and Acquired Immunodeficiency Syndrome. In: Mandell GL, Bennett JE, Dolin R. Principles and Practice of Infectious Diseases. Seventh edition. Churchill Livingstone Elsevier Philadelphia 2010; 1620.
- Frickmann H, Sturm D, Finke EJ: Infektionsrisiken durch Fremdgewebseinsprengung und sexuelle Gewalt im asymmetrischen Konflikt. Wehrmed Mschr 2014; 58: 2 - 9.
- Arastéh K, Arendt G, Bader A et al.: Postexpositionelle Prophylaxe der HIV-Infektion. Deutsch-Österreichische Empfehlungen. Aktualisierung 2007; 4 - 42.
- Low N, Chersich MF, Schmidlin K at al.: Intravaginal practices, bacterial vaginosis, and HIV infection in women: individual participant data meta-analysis. PLoS Med 2011; 8: e1000416.
- Kissinger P, Adamski A: Trichomoniasis and HIV interactions: a review. Sex Transm Infect 2013; 89: 426 - 33.
- Wawer MJ, Gray RH, Sewankambo NK, Serwadda D, et al.: Rates of HIV-1 transmission per coital act, by stage of HIV-1 infection, in Rakai, Uganda. J Infect Dis 2005; 191: 1403 - 1409.
- Underhill K, Montgomery P, Operario D: Sexual abstinence only programmes to prevent HIV infection in high income countries: systematic review. BMJ 2007; 335: 248.
- Underhill K, Operario D, Montgomery P: Abstinence-only programs for HIV infection prevention in high-income countries (Review). Cochrane Database Syst Rev 2007; 4: CD005421.
- Weller S, Davis K: Condom effectiveness in reducing heterosexual HIV transmission. Cochrane Database Syst Rev 2001; 3: CD003255.
- Audebert F, Behrens G, Bogner J, et al.: Deutsch-Österreichische Leitlinien zur postexpositionellen Prophylaxe der HIV-Infektion. 2013; 2 - 26-
- Cardo DM, Culver DH, Ciesielski CA, et al.: A case-control study of HIV seroconversion in health care workers after percutaneous exposure. Centers for Disease Control and Prevention Needlestick Surveillance Group. N Engl J Med 1997; 337: 1485 - 1490.
- Schmiedel S, Hertling S, Degen O, et al.: Postexpositionsprophylaxe bei Gewaltopfern – Das „Hamburger Modell“ als Beispiel für eine interdisziplinäre Verzahnung zur Betreuung von Gewaltopfern. Hamburger Ärzteblatt 2009; 1: 35 - 36.
- Srivastava KK, Fernandez-Larsson R, Zinkus DM, Robinson HL: Human immunodeficiency virus type 1 NL4-3 replication in four T-cell lines: rate and efficiency of entry, a major determinant of permissiveness. J Virol 1991; 65: 3900 - 3902.
- Hofstra LM, Sauvageot N, Albert J, et al.: SPREAD Program. Transmission of HIV Drug Resistance and the Predicted Effect on Current First-line Regimens in Europe. Clin Infect Dis 2016; 62: 655 - 663.
- Salou M, Dagnra AY, Butel C, et al.: High rates of virological failure and drug resistance in perinatally HIV-1-infected children and adolescents receiving lifelong antiretroviral therapy in routine clinics in Togo. JIAS 2016; 19: 20683.
- Mayer KH, Mimiaga MJ, Gelman M, Grasso C: Raltegravir, tenofovir DF, and emtricitabine for postexposure prophylaxis to prevent the sexual transmission of HIV: safety, tolerability, and adherence. J Acquir Immune Defic Syndr 2012; 59: 354 - 359.
- The TenoRes Study Group. Global epidemiology of drug resistance after failure of WHO recommended first-line regimens for adult HIV-1 infection: a multicentre retrospective cohort study. Lancet Infect Dis 2016; 16: 565 - 575.
- Kasang C, Kalluvya S, Majinge C, et al. HIV drug resistance (HIVDR) in antiretroviral therapy-naïve patients in Tanzania not eligible for WHO threshold HIVDR survey is dramatically high. PLoS One 2011; 6: e23091.
- Molina JM, Capitant C, Spire B, et al.: ANRS IPERGAY Study Group. On-Demand Preexposure Prophylaxis in Men at High Risk for HIV-1 Infection. N Engl J Med 2015; 373: 2237 - 2246.
- Frickmann H, Picolin KH, Wiemer D, Potinius M, Hagen RM: Prä-Expositionsprophylaxe (PrEP) gegen HIV – aktueller Forschungsstand und mögliche Relevanz für den Einsatz. Wehrmed Mschr 2012; 56: 15 - 20.
- Ouellet E, Durand M, Guertin JR, LeLorier J, Tremblay CL: Cost effectiveness of ‘on demand’ HIV pre-exposure prophylaxis for non-injection drug-using men who have sex with men in Canada. Can J Infect Dis Med Microbiol 2015; 26: 23 - 29.
- Vollmuth R. Beware of the “Slippery Slope”. Healthcare Professionals between Medical Ethics and Military Duties. Medical Corps International Forum 2016; 2016: 2: 35 - 37.
Manuskriptdaten:
Eingereicht: 27.07.2016
Revidierte Fassung angenommen: 05.09.2016
Zitierweise:
Maaßen W, Gottwald C, Frickmann F: „Wie sag ich’s meinem Kinde?“ – Fallbeispiele gut vertragener HIV-Postexpositionsprophylaxen im tropischen Einsatzgebiet. Wehrmedizinische Monatsschrift 2016; 60(12): 366 - 375.
Citation:
Maassen W, Gottwald C, Frickmann F: „How do I put it?“ - case reports on well tolerated HIV post-exposure prophylaxis on deployment in the tropics. Wehrmedizinische Monatsschrift 2016; 60(12): 366 - 375.
Für die Verfasser:
Oberfeldarzt Priv.-Doz. Dr. Hagen Frickmann
Fachbereich Tropenmedizin am Bernhard-Nocht-Institut, Bundeswehrkrankenhaus Hamburg
Bernhard-Nocht-Straße 74, 20359 HAMBURG
E-Mail: Frickmann@bni-hamburg.de
Eine Deutsche Version des Textes finden Sie hier.
A german version of this article you will find here.
Date: 01/24/2017
Source: Wehrmedizinische Monatsschrift 2016/12