
Article: B. J. MANION, A. MCLAUGHLIN (AUSTRALIA)
Traumatic Brain Injury - A Brief Review of the Acute Care of Head-Injured Patients
In both civilian and military settings, head injury, also referred to as Traumatic Brain Injury (TBI) can be a devastating, highly debilitating and extraordinarily expensive injury. The epidemiology of TBI in the military is difficult to assess accurately.
With improvements in delivery of medical care and protective equipment, soldiers are now surviving injuries they previously would not have survived. This has forced the medical community and society to develop an approach to rehabilitating and accommodating the long-term care needs of more and more soldiers affected by TBI.
The burden of TBI on military personnel is huge. Head and neck injuries, including
severe TBI have been reported in 25% of US service members evacuated from Iraq
and Afghanistan (1,2). Up to 40% of military mortality is attributed to TBI (3,4).
Some researchers have suggested over 300 000 U.S. veterans of the wars in Iraq and
Afghanistan have sustained a mild TBI (4). Official statistics based on electronic medical
records within the U.S. military released by the Department of Defense report 195,547 TBIs were diagnosed in military personnel between 2000-2010. Of this number 2038 were classified as severe; 33 020 were classified as moderate and 150 222 were classified as mild as of November 2010 (1). Research into the epidemiology of TBI in military personnel is a high priority, with the U.S. Senate recently allocating significant funding. Prompt and rational
treatment of the acutely head-injured individual can make a significant difference
to the individual’s subsequent outcomes. Understanding the appropriate
assessment, triage and treatment of personnel suffering a TBI is essential to
achieving the best possible outcome. The pathophysiology, physiology and acute
care of head injury are the main focus of this article.
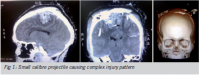
Primary Brain Injury
The injury sustained by the brain at the time of trauma is the primary injury. It
results directly from the trauma and by definition it is irreversible, with prevention
being the only possible intervention. Classification of primary brain injury
based on mechanism of injury identifies three types of TBI; Penetrating; Closed;
and Blast(4,5,6). Single injuries may be a complex combination of these forces.
In penetrating TBI (pTBI) a foreign object penetrates the skull and travels
through the brain parenchyma, damaging neurons, glia and fibre tracts (5). In closed
TBI (cTBI) disruption of brain function can occur from brain motion and deformation
within the cranial vault, resulting in damage to brain parenchyma, blood
vessels and fibre tracts (5). Blast TBI (bTBI) is currently the predominant cause of TBI
in the military (5). An explosive blast wave induces localized particle motion in
the medium it is passing through. The blast wave creates deforming forces, and
the local micromotion of the skull creates a local system of vacuum and pressure
with resultant widespread neurological damage (5).
There may also be tertiary and quaternary blast effects that directly influence
the brain injury. These include impact of material thrown by the blast, the patient
being thrown by the blast, and factors not already specified such as burns (5).
Primary brain injury can be further stratified by severity, based on the patients
Glasgow Coma Scale score, physical signs and symptomatology; and morphology,
which is based on the presence of skull fractures and intracranial lesions (7).
Physiology of Secondary Brain Injury
At the time of the initial injury a cascade of events is set in motion by the initial
insult. This cascade includes, but is not limited to; perfusion abnormalities
leading to ischaemia; haemorrhage, metabolic derangements, multifactorial
oedema; intracranial mass lesions and increased intracranial pressure of several
aetiologies (6,7,8,9,10). This cascade amplifies the initial traumatic injury and
significantly worsens the outcome. The cerebral vasculature has the ability
to maintain a constant amount of blood flow to the brain, largely independent of
systemic blood pressure (11). Auto-regulation is often severely impaired in TBI
patients leading to impaired cerebral blood flow (11). Compounding this, as
intracranial pressure (ICP) increases, cerebral blood flow (CBF) is further compromised.
CBF depends on cerebral perfusion pressure. Cerebral perfusion pressure (CPP)
is the difference between the mean arterial pressure and the ICP (CPP = MAP - ICP)
(11). The critical threshold for ischaemia is a CPP of 50 mmHg (10,11). Rising ICP
leads to decreased brain perfusion, and hence decreased oxygenation, and eventually
ICP will reach levels great enough to cause cessation of arterial flow and the
herniation of regions of brain parenchyma rapidly causing neurological decline and
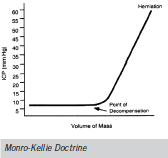
ultimately death (10,11). The Monro-Kellie doctrine is used to describe intracranial pressure dynamics in normal and pathological states (10). The skull provides a set volume to contain CSF, venous and arterial blood, and brain parenchyma. Any increase in the volume
of one of these constituents means a compensatory decrease in volume of the others.
This helps to maintain an ICP within a normal range of 10-15 mmHg. When
this compensatory mechanism is exhausted and a critical intracranial volume
is reached, a point of decompensation is reached with further small
increases in intracranial volume causing an exponential rise in ICP (10,12).
Secondary Brain Injury
Therapeutic strategies are initiated as early as possible after the primary insult in
order to prevent secondary brain injury (3,6,8,9). Lack of rational and timely treatment
can be catastrophic. Adequate oxygenation and maintenance of sufficient
blood pressure to ensure cerebral perfusion are of paramount importance. A single
episode of hypotension in patients with severe TBI is associated with a doubling
of mortality (60% vs 27%) (10). The presence of hypoxia in addition to
hypotension is associated with a mortality of approximately 75% (10). In a study of
100 consecutive patients presenting to a civilian ED with severe TBI, 30% were
hypoxaemic, 13% were hypotensive and 12% had a haematocrit less than 30% (10).
Assessment
Assessment of trauma patients in the military setting can be extremely challenging.
Not only because of the often potentially dangerous environment in which it
must take place, but also because of the high incidence of multiple trauma
patients. In one civilian series of severe TBI patients, more than 50% had additional
major injuries requiring care by multiple specialities (10).
The initial assessment should involve a team approach to history and examination
and follow a rational acute trauma format (3). The following provides an outline:
Patient factors - Demographical/biographical information as necessary
MIST - Mechanism of injury (time, nature of forces involved, concurrent
injuries or fatalities, damage to personal protective equipment/vehicle)
Data logging e.g. accelerometers
Injuries sustained or suspected:
- obvious head/neck/trunk/limb
- obvious distracting injuries
- injuries suspected on mechanism
Signs:
- vital signs
- Glasgow Coma Scale (GCS)
- Pupil size and reactivity
Σ
Primary Survey – Notably airway maintenance with C-spine protection,
breathing and adequate ventilation, restoration of circulation
with control of haemorrhage
Primary and secondary surveys are repeated as necessary if the patient’s clinical
condition changes. It is extremely important to keep accurate and contemporaneous
notes including nature of treatments given. If a head injury is suspected
or encountered it is vital to perform a rapid and directed neurological exam
before the patient is sedated or pharmacologically paralysed. If the clinical situation
allows, serial examinations are to be performed in order to detect any clinical
decline early. When recording the GCS report the patients left and right sides separately.
Following the patients best responses with serial examinations allows
for the detection of deterioration as early as possible (10). Assessment of the GCS at
an early stage will allow determination of the severity of the TBI, and assist with
triage and evacuation decisions (7). It also gives a degree of prediction to the course
of the trauma and the likelihood of other injuries.
- GCS 14-15 -> mild TBI
- GCS 9-13 -> moderate TBI
- GCS 8 or less -> severe TBI
Pupillary examination can be performed quickly with minimal equipment and is
objective (3,10) . Asymmetry between the two pupils in the same patient can be
indicative of severe brain dysfunction (10,13). Mild dilatation of the pupil and a
sluggish pupillary response to light can indicate early uncal herniation (3,10,13).
Care must be taken to rule out primary eye injury in assessing the import of papillary
reactions (10).
Management of mild TBI
Signs and symptomatology can be very subtle and diagnosis often requires specialised
assessment tools. A patient suffering a mild TBI may exhibit a brief loss of
consciousness; disorientation or confusion and amnesia (1,4,5). It is important to
note however, that a patient suffering a mild TBI may be asymptomatic initially
and may not develop symptoms until subjected to further cerebral insults (5).
It is important to remove patients with suspected mild TBI from the field (5).
Three percent of these patients deteriorate rapidly resulting in severe neurologic dysfunction
unless treated early. Up to 18% of mild TBI patients had abnormalities on
initial CT and 5% required surgery (10).
Individuals who sustain a second head injury within 2 weeks of the first are at
risk of developing cerebral oedema. Disruption of cerebral auto-regulation can
lead to cerebral hyperaemia and in the face of a second insult to the brain, this
can occur with extreme rapidity, leading to so-called malignant cerebral oedema.
This scenario is often referred to as the second impact syndrome, and carries a
mortality of up to 100% (10). Triage and transport decisions in combat
scenarios may be complicated by tactical conditions. However, if the situation
allows all personnel with mild TBI should be removed from the field and receive a
CT head scan (10,11). Tools such as the U.S. Department of Defences military
acute concussion evaluation (MACE) and the automated neuropsychological assessment metrics (ANAM) can be used to evaluate a person in whom a concussion is
suspected in the first instance (2), and compare post-injury cognitive function
with pre-deployment cognitive assessment in the case of ANAM (14).
Management of moderate TBI
Patients suffering a moderate head injury will be symptomatic. They may be
confused; aggressive; somnolent or demonstrate focal neurological deficits
(10). Up to 40% of the cases had an abnormal initial CT head scan and 8% required
surgery (10). Further to this, between 10-20% of patients with moderate TBI will
deteriorate to GCS <8 (10). For these reasons patients with moderate TBI are generally
treated aggressively. Protection of the airway is essential and
maintenance of cardiopulmonary stability should be ensured (3,10). All personnel
suffering moderate TBI should be removed from the field with medical support and
airway capabilities. A CT scan of the head is mandatory along with any other investigations
indicated based on concomitant injuries and clinical status.
Patients should be observed in a medical facility capable of dealing with severe
head injuries and have serial neurological assessments. If the patient continues to
improve and follow up CT head shows no surgical mass lesion then a decision
regarding return to action can be addressed by the appropriate chain of
command in due course. If the patient deteriorates, management principles of
severe TBI will be adopted (10).
Management of severe TBI
Mortality rates for patients with severe TBI vary within the literature from 30-
75% (6,7). Prompt diagnosis and treatment is of utmost importance. It is imperative
that cardiopulmonary stabilisation be achieved rapidly in patients with severe
TBI (3,10).
The most important aspect of immediately managing these patients is early
endotracheal intubation and ventilation with 100% oxygen (3,10). Initially
patients should be ventilated to normocarbia (3,10). Hyperventilation may be used
cautiously in patients who demonstrate a worsening GCS or pupillary dilation (10).
Hypotension and hypoxia are the principal causes of deterioration in headinjured
patients (3,6,10,14). Hypotension is not usually due to the brain injury itself,
except in the terminal stages when medullary failure supervenes. Hypotension
is much more often due to hypovolaemia from blood loss, which may be
occult. The clinician must also consider associated spinal injuries, pericardial tamponade
and tension pneumothorax. It is important to note that a hypotensive
patients’ neurological examination is unreliable and may return to near-normal
following resuscitation to euvolaemia (10,15,16).
Once the patients cardiopulmonary status has been stabilised rapid neurological
assessment follows. Avoid using long-acting paralytics and use small repeated doses
of an opiate for pain relief to avoid confounding the neurological assessment (10).
In one series of patients with severe TBI more than 50% had major systemic
injuries, these were: long-bone or pelvic fracture - 32%; maxillary or mandibular
fracture 22%; major chest injury - 23%; abdominal visceral injury - 7%; spinal
injury 2% (3). There is also a strong association between severe brain injury and Cspine
injury. Some of these issues may require evaluation prior to the patient being sent for head CT.
Severely head-injured patients should be evacuated early with full medical support
to a tertiary care facility with ICU and neurosurgical capabilities (10,15,16). The
majority of institutional episodes of hypoxia and hypotension occur during
transfers, and these need to be carefully planned in advance.
Further medical management
Monitoring
Severely head-injured patients need invasive monitoring. All patients should
have their vitals signs continuously monitored (10,17). Glucose and electrolytes
should be monitored as frequently as required to keep them within acceptable
limits. Arterial lines should be employed to provide accurate BP monitoring. Jugular
venous O2 saturations should be monitored if aggressive hyperventilation is
being considered (7,12,17). All patients with severe TBI deemed potentially salvageable
should have intracranial pressure monitoring (12).
ICP assessment and control of intracranial hypertension (IC-HTN)
intracranial hypertension (IC-HTN) After initial resuscitation, the majority
of medical and surgical effort goes into diagnosing and alleviating IC-HTN. Once
the point of decompensation on the intracranial volume - pressure curve is
reached, salvaging the patient becomes exponentially more difficult and neurological
decline and death is imminent (10).
Clinical signs of IC-HTN include; pupillary dilatation (unilateral or bilateral);
asymmetric pupillary reaction to light; decorticate or decerebrate posturing or
progressive deterioration of the neurological exam not attributable to identifiable
extra-cranial factors (3). Cushing’s triad (of hypertension, bradycardia, respiratory
abnormalities) is a late sign, present in ~33% of cases of IC-HTN (18,19).
Intracranial pressure is measured using an intraventricular catheter (IVC) or probe
(3,10).Insertion is an invasive procedure, requiring excellent sterility but minimal
equipment. Potential complications include haemorrhage, which is clinically
significant in 2% of cases, and more likely in a multitrauma or coagulopathic patient.
An external ventricular drain can also be used to rapidly reduce CSF pressure in
acute hydrocephalus. ICP should be kept < 20mmHg in all cases (18).
Additional measures to control ICP include:
Elevate head of bed to 30-45 degrees to decrease ICP by enhancing venous outflow
Avoid hypertension as this will contribute to hyperaemia and increase ICP
Ventilate to normocarbia, as reduced CO2 will cause cerebral vasoconstriction
and reduce cerebral blood flow thereby decreasing intracranial volume. This
should be used with care. Sedation reduces elevated sympathetic
tone and hypertension induced by movement If the general measures listed above fail
to adequately control IC-HTN, the management can include heavy sedation,
hypertonic saline, drainage of CSF, aggressive hyperventilation and mannitol.
Hypothermia is not proven as an effective intervention. Surgical decompression is an
excellent option if available (10).
Mannitol and hypocarbia are useful as short term measures if definitive surgical
care is anticipated (20). Their main use is as a temporizing measure in a patient with
uncal herniation. Hyperventilation can reduce the ICP 25-30% with a PaCO2 of
29mmHg, but can cause a worsening of the CBF and an exacerbation of injury
(20,21). They should be used cautiously, and avoided in volume depleted patients
(10) or those with chest injuries (17). Corticosteroids have no role in the acute management of head trauma.
Prophylactic anti-epileptic drugs
The incidence of early post traumatic seizures (PTS) in patients with severe TBI is
~30%; and that of late PTS in the same patient group is ~10-13%5. Prophylactic
use of anti-epileptic drugs (AEDs) is indicated in patients with severe TBI to reduce
the incidence of early PTS and avoid adverse effects associated with seizures
such as increased ICP, increased BP, metabolic dysfunction and increased oxygen
demand. Patients with acute intracranial mass lesions; open-depressed skull fractures
with parenchymal injury; penetrating injury and seizures within the first 24 hours are all high-risk for early PTS (10).
Continuation of AEDs longer than one week is
generally unhelpful except in
those with penetrating injuries.
Prophylactic AED use does not
reduce the incidence of late PTS (10).
Surgical management
Surgical management of the headinjured patient may be used in the
treatment of skull fractures, IC-HTN, scalp wounds and the evacuation/
decompression of intracranial mass lesions.
Closed depressed skull fractures may be managed surgically or medically. There is
no difference in outcomes in terms of seizure incidence, neurologic dysfunction
or cosmetic appearance (10). Open depressed fractures tend to be managed
surgically if the fragment is depressed more than the thickness of the calvaria (or arbitrarily
>1cm) and if there is evidence of dural penetration, significant intracranial
haematoma, frontal sinus involvement, contamination or gross deformity (10).
Extradural haematomas (EDH) occur in head-injured patients with an incidence of
around 1% and have an overall mortality of 20-55%. Expeditious diagnosis and
treatment can see this drop to 5-10% (3,7,10). Patients with EDH and a GCS <9,
or an estimated volume >30ml should undergo urgent surgical evacuation (10).
Subdural haematomas (SDH) are twice as common as EDH and generally have a
greater mortality. The mortality of SDH ranges from 50-90%, although this figure
is distorted by a more elderly patient demographic. The optimal management is
usually surgical evacuation, recommended within 4 hours.
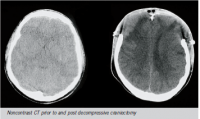
Decompressive craniectomy, although gaining favour with some authors as an
early intervention, is recommended only after medical management has proven
ineffective. Operation can reduce ICP to <20mmHg in 85% of patients (8). Approximately
30% of this pressure drop is due to removal of the bone flap and the remaining
70% to the durotomy (8). Some authors suggest the procedure needs to
occur within 6 hours of the TBI due to the rapidity with which life-threatening cerebral
oedema can occur (8). Others suggest the timing isn’t strictly relevant (10), and
cases should be evaluated on their individual merits. A large civilian trial is currently
assessing the usefulness of this intervention.
Conclusion
Traumatic brain injury continues to be of vast significance in both military and
civilian settings. It is a common and potentially devastating injury with the
potential for long-lasting, far-reaching effects. Blast effects are the predominant
cause of TBI in modern warfare (5). The pathophysiology and epidemiology of TBI
secondary to blast injury are still being investigated.
Approach to the head-injured patient in the field however is relatively standard.
The head-injured patient requires rapid primary assessment and early management
based on well-known and standard guidelines such as EMST/ATLS. Clinicians
must expedite the stabilisation of cardiopulmonary status and avoid secondary
insults such as hypotension and hypoxia. A range of medical and surgical options
exist for the management of secondary brain injury. Ultimately, the medical unit
must rapidly assess and manage the clinical presentation with impeccable application
of first principles. They must decide on the likely course and evacuate the
patient to the health facility best equipped to deal with their needs. Ultimately, accurate
triage, protection of airway and Cspine, and supporting cardiovascular function
are the most critical factors in early management of a head injury (3,10).
Date: 07/12/2011
Source: MCIF 2/11










