
Article
CT-based evaluation of aortic valve dimensions before transcatheter aortic valve implantation: Semi-automatic measurements compared with manual measurements
From the Departments of Radiology1 (Director: Coloneld (MC) Dr S. Waldeck) and Internal Medicine2 (Director: Colonel (MC) Prof Dr C. Bickel), Bundeswehr Central Hospital Koblenz (Commander: Brigadier (MC) Dr J. Brandenstein) in Cooperation with the Bundeswehr Institute for Radiobiology affiliated to Ulm University3, Munich (Director: Colonel (MC) Assistant Professor Dr M. Port), the Institute for Radiology of the Hannover Medical School4 (Director: Prof Dr F. Wacker) and the Central Institute of the Bundeswehr Medical Service Koblenz5 (Director: Colonel (MC) Prof Dr Dr D. Leyk).
Background: Accurate fitting of the prosthesis is crucial for successful transcatheter aortic valve implantation (TAVI) and the prevention of post-interventional adverse events. Therefore, it is essential to obtain accurate aortic valve dimensions of the annulus size and parameters like the dimensions of the ascending aorta and height of the sinus of Valsalva. Reliability of semi-automatic analysis of CT scans as a simple and repeatable procedure to size the aortic annulus was investigated.
Methods: 52 patients who were scheduled for TAVI underwent an ECG-gated multislice CT scan of the aortic valve plane in addition to echocardiographic measurements. Parameters based on the CT scans were then assessed with a standard digital 3D measurement tool. We retrospectively reeva-luated the CT scans with the Vital™Vitrea®fX Advanced 6.2 TAVR-Programme, a semi-automatic 3D-measurement tool. These results were compared with the standard 3D measurement tool as well as with the echocardiographic measurements and the peri-interventional results.
Results: Valve plane area and aortic annulus diameter were significantly overestimated in manually-assessed CT measurements compared to results from the semi-automatic tool. The average aortic annulus diameter differed by > 4 mm in echocardiography compared to results from manual CT measurement and by < 1 mm from semi-automatic 3D-measurement. Concordance between prosthesis size determined by use of the semi-automatic tool and the actual size of the implanted valve was better.
Discussion: Although CT offers a lower interobserver vari-ability compared to transesophageal echocardiographic measurements, this study shows that manual assessment of the aortic valve plane appears error-prone. The incorrectly selected valve plane seems to be most likely the main reason for the systematically overestimated annulus diameter using manual assessment. The semi-automatic measuring method facilitates the multiplanar-wise correct plane level selection from multiple planes. This results in a significantly reduced dispersion width of the semi-automatic measurements.
Conclusions: CT-based assessment of the aortic valve plane using a modern semi-automated reconstruction tool prior to TAVI could provide a more accurate evaluation of the aortic valve dimensions. The use of new reconstruction algorithms - especially for the evaluation of complex traumatic vessel lesions in the case of combat related injuries - could be of interest as well and should be further investigated.
Keywords: TAVI, computed tomography, semi-automatic measurement, aortic valve dimensions, Vital™Vitrea®fX Advanced
Introduction
Aortic valve stenosis is the most common acquired valvular heart disease in elderly patients [1]. Mortality is very high (> 50 % in 2 years) for symptomatic patients with severe aortic stenosis. Up to 33 % of patients who need definite therapy cannot undergo surgery because of several comorbidities [2]. Since the first successful transcatheter aortic valve implantation (TAVI) by CRIBIER et al. in 2002 [3], the number of patients who undergo TAVI has risen dramatically [3, 4]. Previous studies have shown that the clinical outcome after TAVI can be equivalent to the results of conventional surgery [5, 6]. Constant development of new types of prostheses and the improvement of interventional approaches may further improve clinical outcomes. Currently, TAVI is indicated in younger as well as elderly patients, and thus this device is useful for more than palliative care [4]. Nevertheless, TAVI is still associated with a variety of clinical complications. Post-interventional paravalvular leakage is frequent, which sometimes leads to hemodynamic instability, and increases the risk of an unsuccessful intervention [7, 8]. This adverse event, besides -other factors like calcification of the leaflets, seems to be asso-ciated with non-optimal pre-interventional sizing of the prosthesis due to individual anatomical variability [9]. More than 30 % of patients who undergo TAVI show new-onset, high-grade atri-oven-tricular block and need pacemaker implantation [10]. This is probably caused by direct mechanical irritation of the bundle branches and their fascicles when the prosthesis is positioned during the procedure [11]. BLEIZIFFER et al. described post-interventional arrhythmic episodes, and the need for pacemaker therapy may be associated with the implantation of a slightly oversized prosthesis [12].
Therefore, it is crucial to be able to measure the individual aortic annulus size and the optimal prosthesis size before the intervention. At the moment, the choice of the correct prosthesis is based on 2D and 3D echocardiographic measurements [13, 14] as well as on images acquired by multislice computed tomography (MSCT) [15]. Because of recent advancements in the technical development of CT diagnostics, it is already possible to acquire prospectively gated one-shot volume data of the heart during a single cardiac cycle to evaluate the aortic annulus [16]. Until 2-3 years ago, measurements were usually performed with standard 3D reconstruction software. Since then, recently released software for pre-interventional TAVI planning using CT promises to give a semi-automatic identification of the valve plane and a simplified evaluation and finds its way into routine. In this study, CT data sets were reevaluated retrospectively by means of semi-automatic reconstruction software, -Vital™Vitrea®fX Advanced 6.2 TAVR (Vital Images Inc.™, Minnetonka, MN, USA). The results were compared with the original manually-assessed CT results as well as the results obtained by echocardiography. Finally, the resulting parameters of the different tests were compared with the prostheses implanted.
Given the relevance of the optimal prosthesis selection and its impact on clinical outcomes, the purpose of this study was to detect a possible advantage (and quantify it) of the semi-automatic software, as compared with the manual methods currently used. Furthermore, the use of this semi-automatic software might enable interventional cardiologists to make the best possible prosthesis selection.
Methods
52 patients consecutively evaluated for TAVI between January 2012 and December 2013 were included in this retrospective study. All patients suffered from high-grade aortic valve stenosis and were assessed for TAVI in accordance with international guidelines [1, 17] by an interdisciplinary board. Thirty-eight patients were chosen to undergo TAVI. In total, 5 Core--
Valve™-Valves (Medtronic Inc., Minneapolis, MN, USA) were im-planted using a transfemoral access route, and 33 Sapien--
XT™-Systems (Edwards-SAPIEN™, Edwards Inc., USA) were implanted using a transfemoral access route in 28 and a transapical access route in 5. The procedure was successfully performed on all patients.
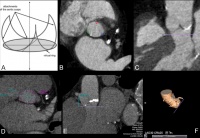 Fig. 1: The aortic annulus plane is a virtual ring, which is derived
from the coronary cusp batches (A, adapted and modifi ed from
Achenbach et al., 2012). In vivo, this plane has a round to ovoid
shape that depends on the individual as well as the phase of the
cardiac cycle (B, C, D). The distances between this plane and the
coronary artery must be measured orthogonally to ensure correct and
secure implantation procedures (E). A three-dimensional reconstruc-
tion allows the orientation in space and provides information for
angulations needed during the intervention (F).
Fig. 1: The aortic annulus plane is a virtual ring, which is derived
from the coronary cusp batches (A, adapted and modifi ed from
Achenbach et al., 2012). In vivo, this plane has a round to ovoid
shape that depends on the individual as well as the phase of the
cardiac cycle (B, C, D). The distances between this plane and the
coronary artery must be measured orthogonally to ensure correct and
secure implantation procedures (E). A three-dimensional reconstruc-
tion allows the orientation in space and provides information for
angulations needed during the intervention (F).
MSCT was performed prior to the intervention to measure the aortic annulus diameter and to evaluate the angiographic access route as well as to detect relevant vessel stenosis or other pathological findings that would lead to a problematic intervention.Highly-experienced cardiologists (> 15 years) performed the echocardiographic assessment of the aortic valve annulus di-ame-ter and the individual anatomic variations of the coronary vessels by 2D and 3D transthoracic and transesophageal echocardio-graphy. CT scans were performed with a modern multislice scanner (Siemens Somatom Sensation 64, Siemens Healthcare, Forchheim, Germany) that had a 4 cm detector range and 64 rows. A prospectively ECG-gated scan of the heart with focus on the aortic valve plane, and a helical scan of the thorax and abdomen were included in the standard protocol. Both scans were performed with an iodine contrast agent (Xenetix 350, Guerbert AG, Villepinte, France) using the bolus-triggering technique (CAREBolus™, Siemens Medical, München, Germany) within the arterial contrast-enhanced phase. Contrast agent was injected intravenously through a secure line at an average flow rate of 4 ml/s. The dose was adjusted for each patient. The images generated were obtained with the filtered back-projection technique (aortic valve annulus plane: medium smooth kernel B30f, slice 0.75 mm, pitch 0.6 mm; angiographic scan: smooth kernel B20f, slice 0.75 mm, pitch 0.6 mm). The initial CT scans were assessed and evaluated independently by two radiologists with special expertise in the field of cardiovascular imaging (> 5 years). For the retrospective assessment of the acquired data, slices of 1 mm thickness within a medium smooth kernel were used in the axial, coronal and sagittal orientation. Image interpretation before TAVI was based on the general recommendations for the assessment of CT data [15]. After identification of the virtual aortic annulus plane, the following parameters were measured using commercially-available 3D reconstruction software (Syngo™ VX91B, Siemens Medical, München): aortic annulus area and average diameter, distance between the ostium of the left/right coronary artery and the aortic annulus, and the minimal diameter of the ascending thoracic aorta. Valid results of the mean diameter were drawn mathematically from the aortic annulus area, according to recommendations in the current literature [18].
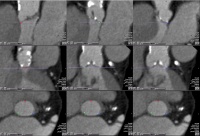 Fig. 2: The fi gure shows marked batches of the aortic cusps in three
standard spatial planes in vertical columns. The right-coronary cusp
(red) is marked in the left column, the non-coronary cusp (green) is
marked the middle column and the left coronary cusp (blue) is
marked in the right column. The marking of the cusp batches was
performed in the same order. Particularly noteworthy is the possibility
of controlling the correct labeling of the batches in three spatial
planes. In every spatial view, at any time during the process,
readjustment is possible. After inserting the three markers in the
orthogonal cut of the inlaid aortic annulus valve plane, these markers
will also appear in the sagittal and coronal views.
Fig. 2: The fi gure shows marked batches of the aortic cusps in three
standard spatial planes in vertical columns. The right-coronary cusp
(red) is marked in the left column, the non-coronary cusp (green) is
marked the middle column and the left coronary cusp (blue) is
marked in the right column. The marking of the cusp batches was
performed in the same order. Particularly noteworthy is the possibility
of controlling the correct labeling of the batches in three spatial
planes. In every spatial view, at any time during the process,
readjustment is possible. After inserting the three markers in the
orthogonal cut of the inlaid aortic annulus valve plane, these markers
will also appear in the sagittal and coronal views.
In addition, the study focused on the assessment of common pathological findings, particularly on the consideration of possible contraindications for interventional aortic valve replacement, like high-grade stenosis of the angiographic access route or aortic aneurysms and dissections. Any individual anatomical variations were registered. The subsequent examination used the same basic data, but was independently assessed by two radiologists who were blinded to the original results and to each other’s results. The difference between the first and second CT assessments was the use of reconstruction software for image post-processing and analysis. Vital™Vitrea®fX Advanced 6.2 TAVR software was used for semi-automatic identification as well as measurement of the area of the aortic annulus valve plane. The origins of the aortic cusps were identified and marked, starting with the right coronary cusp and rotating clockwise thereafter. After validation of the correct identification of these structures in three standard spatial planes, the program automatically set an orthogonal valve plane (figure 1 and 2). Subsequently, the semi-automatic measurement of the areas and distances was performed and required only sporadic manual adjustment (figure 1).
Statistics:
McNemar’s test was used for the comparison of dichotomous matching odds between the annulus plane size that was determined from the CT scans and the aortic prosthesis that was actually implanted. The results from the manual and semi-automatic CT measurements of the distance from the coronary arteries to the aortic annulus valve plane and the aortic annulus diameter were compared using the Wilcoxon signed-rank test. The comparison of methods for the evaluation of aortic annulus diameter was shown graphically by Bland-Altman plots. Continuous variables were reported as the mean ± standard deviation (SD), except for the NYHA functional class, which was reported as the median. A p-value < 0.05 was defined as significant for all tests. SPSS® 20 (IBM, Armonk, NY) was used for all statistical analyses.
Results
Patients
A total of 52 patients were included in the study, and CT scans were evaluated prior to TAVI. The average age of the patients was 77.7 years (61 - 90 years). The group showed a very high degree of morbidity, as indicated by individual EuroScores > 20 %. Most patients were symptomatic (NYHA class 3 ± 0.45), and signs of high-grade aortic stenosis were found (mean gradient 52.0 ± 15.9 mmHg). Thirty-eight patients were selected for TAVI and underwent successful implantation.
Measurements
Measurements showed variable results for the mean aortic annulus diameter assessed by the different techniques (Figure 3). On average, the smallest diameter was obtained by echocardiography (24.05 ± 2.27 mm). With the semi-automatic CT assessment, the diameter was only slightly larger (25.03 ± 2.79 mm). The largest aortic annulus diameter was obtained by manual CT assessment (28.53 ± 4.64 mm).
Comparison of methods
Manual versus semi-automatic analysis of CT scans
The area and diameter of the aortic annulus measured by manual analysis of the CT scans differed significantly from the results obtained by reevaluation of the CT scans using the semi-automatic software (p < 0.001). The results of the mean diameter show differences of > 3.5 mm on average (manually assessed: 28.53 ± 4.64 mm vs. semi-automatically assessed: 25.03 ± 2.79 mm). In more than 84% of 44 patients, a smaller aortic annulus diameter was obtained using the semi-automatic method compared with the manual method (Figure 3 and 4). The assessment of the distance between the ostium of the left coronary artery to the valve plane revealed no significant differences (manual assessment: 13.68 ± 3.12 mm vs. semi-automatic assessment: 13.79 ± 3.03 mm; p = 0.75). However, a significantly greater distance between the aortic valve plane to the ostium of the right coronary artery was obtained using the semi-automatic measurement method (manually assessment: 14.29 ± 4.21 mm vs. semi-automatic assessment: 15.63 ± 4.10 mm; p = 0.026).
CT versus Echocardiography
In comparison to the results obtained by semi-automatic or manual assessment of the CT scans, the echocardiographic measurements performed before the interventions reveal significantly divergent results. On average, the difference between the semi-automatic and echocardiographic measurements did not exceed 1 mm (echocardiography: 24.05 ± 2.27 mm vs. semi-automatically assessed CT: 25.03 ± 2.79 mm). Notably, the difference between the diameter of the aortic annulus obtained by manual CT assessment and by echocardiography was > 4 mm (Figure 3 and 4).
CT versus implanted prosthesis
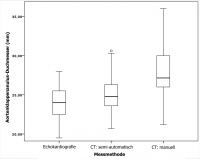 Fig. 3: This fi gure shows box and whisker plots of the aortic annulus
diameter based on three different methods. The middle line in each
box shows the median, the top and bottom of each box shows the
interquartile range and the whiskers show the deviation-free range.
The smallest diameters were obtained by echocardiography. Com-
pared with the other two measurement methods, signifi cantly higher
diameters were obtained by manual CT evaluation (echocardiogra-
phy: mean 24.05 mm, range 19.50 – 28.00 mm, 95% CI 23.31 –
24.07 mm; semi-automatic CT assessment: mean 25.03 mm, range
19.00 – 34.40 mm, 95% CI 24.26 – 25.81 mm; manual CT assess-
ment: mean 28.53 mm, range 21.20 – 48.00 mm, 95% CI 27.24 –
29.83 mm).
Fig. 3: This fi gure shows box and whisker plots of the aortic annulus
diameter based on three different methods. The middle line in each
box shows the median, the top and bottom of each box shows the
interquartile range and the whiskers show the deviation-free range.
The smallest diameters were obtained by echocardiography. Com-
pared with the other two measurement methods, signifi cantly higher
diameters were obtained by manual CT evaluation (echocardiogra-
phy: mean 24.05 mm, range 19.50 – 28.00 mm, 95% CI 23.31 –
24.07 mm; semi-automatic CT assessment: mean 25.03 mm, range
19.00 – 34.40 mm, 95% CI 24.26 – 25.81 mm; manual CT assess-
ment: mean 28.53 mm, range 21.20 – 48.00 mm, 95% CI 27.24 –
29.83 mm).
Hypothetically simply basing the choice of the implanted prosthesis on the initial manual analysis of the CT scans according to the individual manufactures recommendations would have led to the implantation of another type of prosthesis in many cases. The actual size of the prosthesis selected based on pre- and peri-interventional multimodal assessment resulted in the implantation of a prosthesis that differed considerably from the one that would have been selected based solely on the original manual assessment of the CT scans. Only 38 % of the originally determined prostheses, based on the initial CT results, were consistent with the valves that were successfully implanted. Based on reassessment with the semi-automatic algorithm, 79 % of the results were consistent with the implanted valves. These results could be a first hint towards a slightly better concordance between the valve sizes determined by semi-automatic analysis of the CT scans and the prostheses that were ultimately implanted (Figure 5).
Discussion
Advantages and disadvantages of competing methods
Although there is no ‘gold standard’ for the evaluation of the aortic annulus diameter, transesophageal echocardiographic measurement is usually preferred according to the literature [13, 14].
However, CT offers a low interobserver variability and hypothetically should yield at least equivalent results. In addition, anatomical parameters can be determined such as arterial kinking and pathological arterial deviations such as the degree of calcification [15]. In particular, the use of standardized measurement procedures should lead to consistent measurement results. Consequently, a more reliable evaluation of the CT scans before TAVI seems to be of utmost importance for optimal prosthesis selection and to reduce complications associated with the intervention.
 Fig. 4: Bland-Altman plots showing the agreement between two different measurement tools and a potential systematic error in measurement.
The difference in aortic annulus diameter measured by two methods in each patient is plotted against the average of the two methods in each
graph. The middle auxiliary line shows the mean of the difference distribution, whereas the upper and lower auxiliary lines show ± 1.96 times the
standard deviation of the difference distribution. Plot (A) shows the differences between the measurements obtained by the semi-automatic and
manual analysis of the CT scans. Plot (B) shows the differences between the measurements obtained by echocardiography and the manual
method. Differences between the measurements obtained by echocardiography and the semi-automatic method are shown in plot (C). Note the
large range and signifi cant difference between the means when the CT measurement methods are compared with each other (A), and the manual
measurements are compared with the echocardiographic measurements (B). In contrast, the echocardiographic measurements differ only slightly
from the semiautomatic CT measurements (C).
Fig. 4: Bland-Altman plots showing the agreement between two different measurement tools and a potential systematic error in measurement.
The difference in aortic annulus diameter measured by two methods in each patient is plotted against the average of the two methods in each
graph. The middle auxiliary line shows the mean of the difference distribution, whereas the upper and lower auxiliary lines show ± 1.96 times the
standard deviation of the difference distribution. Plot (A) shows the differences between the measurements obtained by the semi-automatic and
manual analysis of the CT scans. Plot (B) shows the differences between the measurements obtained by echocardiography and the manual
method. Differences between the measurements obtained by echocardiography and the semi-automatic method are shown in plot (C). Note the
large range and signifi cant difference between the means when the CT measurement methods are compared with each other (A), and the manual
measurements are compared with the echocardiographic measurements (B). In contrast, the echocardiographic measurements differ only slightly
from the semiautomatic CT measurements (C).
This study shows that manual assessment of the aortic valve plane with exact positioning of the origins of the aortic valve cusps appears error-prone, in spite of the benefits of the tech-nology applied. The incorrectly selected valve plane seems to be most likely the main reason for the systematically overestimated annulus diameter using manual assessment. The choice of an implanted prosthesis solely based on this assessment would therefore lead to the implantation of an oversized valve. Besides an aggravated risk of a potentially fatal perforation, there is also the risk of mechanical irritation of the bundle branches and their fascicles resulting in high-grade atrioventricular block [11, 12]. The semi-automatic measuring method facilitates the multiplanar-wise correct plane level selection from multiple planes, and it allows the exact positioning of the virtual valve plane in predetermined systematic steps. This results in a significantly reduced dispersion width of the semi-automatic measurements.
In a recently published study, VAN LINDEN et al. were able to demonstrate that a mainly automated measurement with different software (Syngo® AorticValveGuide®, Siemens Healthcare, Forchheim, Germany) provided reliable results for the assessment of the aortic annulus compared with a specialized semi-automatic measurement tool (3mensio® Medical Imaging BV, the Netherlands) [19].
Although the distances between the coronary ostia and the aortic annulus plane differed significantly between the two CT measurement methods used in this study, this had no negative effect on the choice of the prosthesis, since the distances obtained by both measurement methods were within the manufacturer’s recommended parameters. The significant difference in measuring the distances to the right coronary artery was notable, in contrast to a similar measured distance to the left coronary artery. The divergent distance measurements from the valve plane to the right coronary artery are most likely due to the systematic incorrect positioning of the right-coronary cusp and the incorrect determination of the aortic annulus plane. In our experience, it is harder to identify the right coronary cusp than the non-coronary or left coronary cusp. We did not see any hemodynamically relevant obstruction of the coronary ostia in our patients during the intervention.
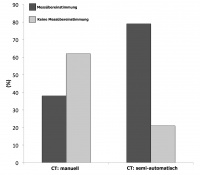 Fig. 5: Agreement between the two different CT measurements and
the implanted aortic valve size. This fi gure illustrates the difference
between the prosthesis sizes based solely on the manual or semi-auto-
matic CT measurements and the aortic valves that were implanted. It
shows greater validity of the semi-automatic measurement technique.
The prosthesis sizes determined by the semi-automatic method
showed better agreement (p=0.001) with the valve sizes that were
ultimately implanted. The agreement between the size of the valve
that was implanted and the prosthesis size based solely on CT
measurement increased by 41 %.
Fig. 5: Agreement between the two different CT measurements and
the implanted aortic valve size. This fi gure illustrates the difference
between the prosthesis sizes based solely on the manual or semi-auto-
matic CT measurements and the aortic valves that were implanted. It
shows greater validity of the semi-automatic measurement technique.
The prosthesis sizes determined by the semi-automatic method
showed better agreement (p=0.001) with the valve sizes that were
ultimately implanted. The agreement between the size of the valve
that was implanted and the prosthesis size based solely on CT
measurement increased by 41 %.
The time required for a complete CT assessment before TAVI was subjectively reduced when the semi-automatic method was used. This promises a better implementation of this method in clinical practice. In rare cases, especially with poor quality data or non-optimal contrast-enhanced images, it was necessary to manually adjust the automatic measurements of the aortic annulus dimensions. This is in accordance with the recently published observations made by LOU et al. [20]. The question remains whether new generations of software and faster acquisition times in new generations of CT scanners can provide a more reliable measurement. The results obtained with the semi-automatic measurement corresponded well with the results obtained by echocardiography. The semi-automatic measurements performed retrospectively demonstrated a high degree of concordance with the implanted valve sizes and therefore showed a higher degree of predictability. The comparison between the aortic annulus diameter determined by echocardiography and semi-automatic CT assessment showed an average difference of only about 0.1 cm. Taking into account the known systematic measurement error of echocardiography, the annulus diameter is usually overestimated by about 0.1 cm, and thus the difference disappears almost completely [21]. WATANABE et al. recently described similar results when testing another commercially-available, automatic measurement tool (3mensio®, Medical Imaging BV, the Netherlands). With the implantation of a self-expanding valve, paravalvular leaks could also be predicted more accurately than with manual measurements [22].
When the conditions for echocardiography are not ideal, such as in obese patients or patients with emphysema or when patients are not compliant, the echocardiographic measurements can be inaccurate [23]. In these cases, measurements from CT scans are crucial in order to select the correct prosthesis. Even in a perfect measurement environment, relevant differences in results based on CT and echocardiography should lead to reevaluation of the results before the valve is implanted. Prosthesis selection based exclusively on manual CT measurements should be avoided.
Impact on choice of prosthesis
The commonly used prosthesis models show gradual differences in sizes of about 3 mm depending on the manufacturer [24]. Since this study found a systematic measurement error of about 5 mm using the manual method to determine the aortic annulus diameter, there is the obvious danger of improper prosthesis selection. Thus, a generally reduced risk of adverse events during and after the intervention should reduce complications associated with the selection of the wrong prosthesis (e.g. arrhythmic episodes or paravalvular leakage). Particularly when taking into consideration the currently changing TAVI indications towards a younger patient population, this seems to be of imminent importance.
Potential value for traumatic vessel injuries
MSCT with an implemented 3D reconstruction algorithm is already standard technique in the assessment of different traumatic injuries [25]. In addition, CT-angiography has already proven to be suitable in the diagnosis of vascular trauma in soldiers [26] as it can help to find occult vessel injuries from combat injuries [27]. New reconstruction tools could provide faster and more accurate assessment of traumatic- and combat vessel injuries and should therefore be investigated further.
Limitations
The validity of the study is limited by several factors. The enrollment of patients at a single center and the sample size limits the assessment. This leads to stochastic effects, and thus additional studies are required with a larger sample size and a longer duration of follow-up. With these new studies, examiner-dependent bias could also be avoided. A high incidence of tachycardia in the population in need of TAVI makes an ECG-gated CT scan impossible in several cases resulting in the exclusion of many patients. However, new-generation CT-scanners promise a higher tolerance when it comes to tachycardia and arrhythmic events, and their use could lead to an expansion of the population that can be scanned properly.
Conclusions
This retrospective study showed that CT evaluation of the aortic annulus using a semi-automatic measurement tool, like the Vital™Vitrea®fX Advanced, produced significantly more accurate results than measurements based on manual assessment of CT scans. The results obtained by semi-automatic assessment showed a better concordance with the echocardiographic data and the implanted valve sizes. The semi-automatic measurement method seems to be a more valid and more inter-observer independent way to evaluate the aortic annulus than manual measurement. In particular, a multimodal evaluation of each patient that includes echocardiography should optimize the measurement results. Moreover, the realization of measurement differences among various modalities and the need for measurement reevaluation may reduce the complications post-interventional. CT based reconstruction tools potentially can provide an optimized assessement of combat related vessel injuries and should be investigated in the future.
Key Message
- Computed tomography is an elaborate method for the pre-interventional workup of patients scheduled for transcatheter aortic valve implantation.
- Appropriate fitting of the prosthesis is crucial for a good clinical outcome.
- A new semi-automatic measurement tool for the accurate assessment of different parameters needed for the intervention was retrospectively evaluated.
- Analysis showed good results in comparison to the manual measurements.
- New reconstruction tools potentially can provide faster and more accurate assessment of combat related vessel injuries.
References
- Vahanian A, Baumgartner H, Bax J, et al.: Guidelines on the management of valvular heart disease: The Task Force on the Management of Valvular Heart Disease of the European Society of Cardiology. Eur Heart J 2007; 28: 230 - 268.
- Lung B, Cachier A, Baron G, et al.: Decision-making in elderly patients with severe aortic stenosis: why are so many denied surgery? Eur Heart J 2005; 26: 2714 - 2720.
- Cribier A, Eltchaninoff H, Bash A, et al.: Percutaneous trans- catheter implantation of an aortic valve prosthesis for calcific aortic stenosis: first human case description. Circulation 2002; 106: 3006-3008.
- Cribier A: Development of transcatheter aortic valve implantation (TAVI): a 20-year odyssey. Arch Cardiovasc Dis 2012; 105(3): 146 - 152.
- Kodali SK, Williams MR, Smith CR, et al.: For PARTNER Trial Investigators. Two-year outcomes after transcatheter or surgical aortic valve replacement. N Engl J Med 2012; 366: 1686 - 1695.
- Smith CR, Leon MB, Mack MJ, et al.; Transcatheter versus surgical aortic-valve replacement in high-risk patients. N Engl J Med 2011; 364: 2187 - 2198
- Rallidis LS, Moyssakis IE, Ikonomidis I et al.: Natural history of early aortic paraprosthetic regurgitation: a five-year follow-up. Am Heart J 1999; 138: 351 - 357.
- Sherif MA, Abdel-Wahab M, Stocker B: Anatomic and procedural predictors of paravalvular aortic regurgitation after implantation of the Medtronic CoreValve bioprosthesis. J Am Coll Cardiol 2010; 56: 1623 - 1629
- Détaint D, Lepage L, Himbert D, et al.: Determinants of significant paravalvular regurgitation after transcatheter aortic valve: implantation impact of device and annulus discongruence. J Am Coll Cardiol Intv 2009; 2: 821 - 827.
- Eltchaninoff H, Prat A, Gilard M, et al.: Transcatheter aortic valve implantation: early results of the FRANCE (FRench Aortic National CoreValve and Edwards) Registry. Eur Heart J 2011; 32 :191 - 197.
- Jilaihawi H, Chin D, Vasa-Nicotera M: Predictors for permanent pacemaker requirement after transcatheter aortic valve implantation with the CoreValve bioprosthesis. Am Heart J 2009; 157: 860 - 866.
- Bleiziffer S, Ruge H, Horer J: Predictors for new-onset complete heart block after transcatheter aortic valve implantation. J Am Coll Cardiol Intv 2010; 3: 524 - 530.
- Messika-Zeitoun D, Serfaty JM, Brochet E. Multimodal assessment of the aortic annulus diameter: implications for transcatheter aortic valve implantation. J Am Coll Cardiol 2010; 55: 186 - 194.
- Vahanian A, Alfieri O, Al-Attar N, et al.: Transcatheter valve implantation for patients with aortic stenosis: a position statement from the European Association of Cardio Thoracic Surgery (EACTS) and the European Society of Cardiology (ESC), in collaboration with the European Association of Percutaneous Cardiovascular Interventions (EAPCI). Eur Heart J 2008; 29: 1463 - 1470.
- Achenbach S, Delgado V, Hausleiter J, et al.: SCCT expert consensus document on computed tomography imaging before transcatheter aortic valve implantation (TAVI) / transcatheter aortic valve replacement (TAVR). J Cardiovasc Comput Tomogr 2012; 6: 366 - 380.
- Lehmkuhl L, Foldyna B, Von Aspern K et al.: Inter-individual variance and cardiac cycle dependency of aortic root dimensions and shape as assessed by ECG-gated multi-slice com- puted tomography in patients with severe aortic stenosis prior to transcatheter aortic valve implantation: is it crucial for correct sizing? Int J Cardiovasc Imaging 2013; 29:693 - 703.
- Holmes DR, Mack MJ, Kaul S, et al.: ACCF/AATS/SCAI/STS expert consensus document on transcatheter aortic valve replacement: developed in collaboration with the American Heart Association, American Society of Echocardiography, European Association for Cardio-Thoracic Surgery, Heart Failure Society of America, Mended Hearts, Society of Cardiovascular Anesthesiologists, Society of Cardiovascular Computed Tomography, and Society for Cardiovascular Magnetic Resonance. Ann Thorac Surg 2012; 93: 1340 - 1345.
- Gurvitch R, Webb JG, Yuan R, et al.: Aortic annulus diameter determination by multidetector computed tomography: reproducibility, applicability, and implications for transcatheter aortic valve implantation. JACC Cardiovasc Interv 2011; 4: 1235 - 1245.
- Van Linden A, Kempfert J, Blumenstein J, et al.: Manual versus automatic detection of aortic annulus plane in a computed tomography scan for transcatheter aortic valve implantation screening. Eur J Cardiothorac Surg 2014; 46(2): 207 - 212.
- Lou J, Obuchowski NA, Krishnaswamy A, et al.: Manual, semiautomated, and fully automated measurement of the aortic annulus for planning of transcatheter aortic valve replacement (TAVR/TAVI): Analysis of interchangeability. J Cardiovasc Comput Tomogr 2014; 14:1934 - 5925.
- Ng AC, Delgado V, van der Kley F: Comparison of aortic root dimensions and geometries before and after transcatheter aortic valve implantation by 2- and 3-dimensional transesophageal echocardiography and multislice computed tomography. Circ Cardiovasc Imaging 2010; 3: 94 - 102
- Watanabe Y, Morice MC, Bouvier E, et al.: Automated 3-dimensional aortic annular assessment by multidetector computed tomography in transcatheter aortic valve implantation. JACC Cardiovasc Interv 2013; 9: 955 - 964.
- Buck T, Breithardt OA, Faber L, et al.: Manual zur Indikation und Durchführung der Echokardiographie. Clin Res Cardiol Supl 2009; 4(1): 3 - 51.
- Al-Lamee R, Godino C, Colombo A: Transcatheter aortic valve implantation: current principles of patient and technique selection and future perspectives. Circ Cardiovasc Interv 2011; 4: 387 - 95.
- Anderson SW, Lucey BC, Varghese JC, Soto JA: Sixty-four multi-detector row computed tomography in multitrauma patient imaging: early experience. Current problems in diagnostic radiology 2006; 35(5): 188 - 198.
- White PW, Gillespie DL, Feurstein I, et al.: Sixty-four slice multidetector computed tomographic angiography in the evaluation of vascular trauma. J Trauma Acute Care Surg 2010; 68(1): 96 - 102.
- Watchorn J, Miles R, Moore N: The role of CT angiography in military trauma. Clinical radiology 2013; 68(1): 39 - 46.
Disclosure of potential conflicts of interest:
All authors indicate no potential conflicts of interest.
Citation:
Becker B, Nestler K, von Falck C, Veit D, Joeckel J, Waldeck S: CT-based evaluation of aortic valve dimensions before transcatheter aortic valve implantation: Semi-automatic measurements compared with manual measurements. Wehrmedizinische Monatsschrift 2017; 61(4): 74 - 80.
Reference address for authors:
Major (MC) Dr Benjamin V. Becker
Bundeswehr Institute of Radiobiology affiliated to Ulm University, Munich, Germany
Neuherbergstrasse 11, 80937 Munich, Germany
E-Mail: [email protected]
Date: 05/04/2017
Source: Wehrmedizinische Monatsschrift 2017/4










